Algae-resistant agent that meets green standards
Categories: Homepage Recommended News
Categories: Technical exchange
Time:2024-03-27 16:27
In humid and wet southern climates, green algae or other plants are often found growing on the surface of the emulsion paint film on the sunny side of exterior walls. This is because algae and other green plants mainly use photosynthesis to generate energy, and then adhere to the paint film and reproduce in large quantities, causing the waterproofness of the paint film to decrease, adhesion to deteriorate, losing its decorative and protective functions, and reducing the service life of the paint film.
In order to kill algae, we need to add algicides to the paint. Diuron is the most commonly used algicidal active substance in paints, but it is prohibited in the green standard GB/T 35602-2017 for paints; GB 18582-2020 In the updated version, diuron will be restricted, so terbutryn, which also has high-efficiency algaecidal performance, has attracted the attention of various paint manufacturers.
Terbutryn prevents algae growth by inhibiting algal photosynthesis, but its specific mechanism of action is generally not well known in the paint industry. Therefore, this article aims to briefly describe the process of photosynthesis and the specific mechanism of the algaecide terbutryn for your reference.
1. Photosynthesis
All organisms capable of oxygenic photosynthesis have two photosystems: Photosystem I (PSI) and Photosystem II (PSII). They convert light energy into chemical energy by excitation and electron transfer through a series of molecules in the thylakoid membrane, which is a very complex process. This is a very complex process. In this process, water photolysis produces oxygen, which is released into the atmosphere. Finally, the electron acceptor NADP+ is reduced to NADPH, and ATP synthase uses the H+ concentration gradient to synthesize ATP from ADP and phosphate, completing the conversion of light energy to chemical energy.
This light-driven electron transport chain is continuous in the presence of light. It involves a series of chemical reactions, such as light absorption, energy conversion, and electron transfer; however, this electron transport chain is linear, meaning that if any one link is broken, the entire chain will be broken, thus preventing the photosynthetic reaction from proceeding.
2. Plastoquinone
There are a series of key enzymes or protein molecules in the photosystem. Today's protagonist is plastoquinone, because it is closely related to the mechanism of action of terbutryn. Plastoquinone is a very crucial transmembrane electron carrier molecule in PS II. Its molecular structure is shown in the figure below:
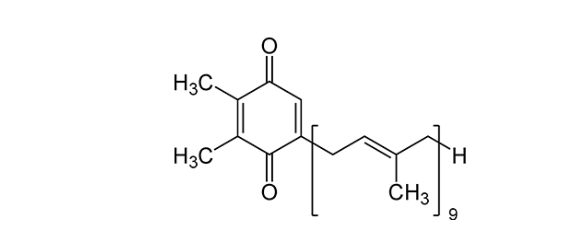
Plastoquinone has two specific binding sites in Photosystem II, called QA and QB. The binding of plastoquinone at QA is very tight, while at QB it is relatively loose and easy to leave. QA only transfers one electron at a time, so before QB can accept two protons and be replaced by another plastoquinone molecule, it must transfer an electron to QB twice.
3. The mechanism of action of terbutryn
The algicidal mechanisms of different algicides vary. Numerous studies have shown that terbutryn mainly competitively inhibits the binding of plastoquinone to the QB site in Photosystem II, thereby interrupting the electron transport chain, hindering the conversion of light energy to chemical energy, and ultimately leading to algal death. So how does terbutryn achieve the inhibition of the binding of plastoquinone to the QB site? The QB binding pocket of PS II contains several key amino acid residues. Plastoquinone forms a hydrogen bond with His215 here. This binding is relatively loose, so it can leave freely after receiving two electrons.
The binding mode of the terbutryn molecule in the QB binding pocket, resolved at 3.2 Å resolution, shows that the terbutryn molecule forms stable hydrogen bonds with key amino acids through three nitrogen atoms on one side. Its N5 can form a hydrogen bond with the backbone amino group of Phe-265A (hydrogen bond distance 3.1 Å); the ethylamino N can form a hydrogen bond with the γ-oxygen atom of Ser-264A (hydrogen bond distance 3.3 Å), and can also form a hydrogen bond with the backbone carbonyl oxygen atom of Phe-265A (hydrogen bond distance 3.2 Å). The tertiary butylamino N forms a hydrogen bond with the backbone carbonyl oxygen atom of Ala-263A (hydrogen bond distance 4.3 Å). These hydrogen bonding interactions can securely anchor the terbutryn molecule in the QB binding pocket, thereby shielding the binding of plastoquinone at the QB site. It is through this mechanism that terbutryn achieves its highly effective algaecidal function, making it widely used in exterior wall coatings.
4. ACTICIDE® SR 2044
ACTICIDE® SR 2044 is a terbutryn-based algaecide specifically for exterior wall coatings. It has a highly effective broad-spectrum algaecidal function. At the same time, it has good system compatibility and durability, making it an ideal choice for algae prevention on exterior walls in high-temperature and high-humidity regions.


